Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O)
O) ]
] with nitrate ions by using density functional theory calculation
with nitrate ions by using density functional theory calculationKato, Akane*; Kaneko, Masashi; Nakashima, Satoru*
RSC Advances (Internet), 10(41), p.24434 - 24443, 2020/06
Times Cited Count:6 Percentile:30.44(Chemistry, Multidisciplinary)Complexation reactions of ruthenium-nitrosyl complexes in HNO solution were investigated by density functional theory (DFT) calculations in order to predict the stability of Ru species in high-level radioactive liquid waste (HLLW) solution. Equilibrium structure of [Ru(NO)(NO
solution were investigated by density functional theory (DFT) calculations in order to predict the stability of Ru species in high-level radioactive liquid waste (HLLW) solution. Equilibrium structure of [Ru(NO)(NO )
) (H
(H O)
O) ] obtained by DFT calculations reproduced the experimental Ru-ligands bond lengths and IR frequencies reported previously. Comparison of the Gibbs energies among the geometrical isomers revealed that the complexation reactions of the ruthenium-nitrosyl complexes with NO
] obtained by DFT calculations reproduced the experimental Ru-ligands bond lengths and IR frequencies reported previously. Comparison of the Gibbs energies among the geometrical isomers revealed that the complexation reactions of the ruthenium-nitrosyl complexes with NO
 proceed via the NO
proceed via the NO
 coordination to the equatorial plane toward the Ru-NO axis. We also estimated Gibbs energy differences on the stepwise complexation reactions to succeed in reproducing the fraction of Ru-NO species in 6 M HNO
coordination to the equatorial plane toward the Ru-NO axis. We also estimated Gibbs energy differences on the stepwise complexation reactions to succeed in reproducing the fraction of Ru-NO species in 6 M HNO solution, such as in HLLW, by considering the association energy between the Ru-NO species and the substituting ligands. Electron density analyses of the complexes indicated that the strength of the Ru-ligands coordination bonds depends on the stability of the Ru species and the Ru complex without NO
solution, such as in HLLW, by considering the association energy between the Ru-NO species and the substituting ligands. Electron density analyses of the complexes indicated that the strength of the Ru-ligands coordination bonds depends on the stability of the Ru species and the Ru complex without NO
 at the axial position is more stable than that wit NO
at the axial position is more stable than that wit NO
 , which might attribute to the difference in the trans influence between H
, which might attribute to the difference in the trans influence between H O and NO
O and NO
 . Finally, we demonstrated the complexation kinetics in the reactions
. Finally, we demonstrated the complexation kinetics in the reactions 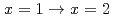 . The present study is expected to enable us to model the precise complexation reactions of platinum-group metals in HNO
. The present study is expected to enable us to model the precise complexation reactions of platinum-group metals in HNO solution.
solution.
Yamashita, Yuji*; Saito, Takumi
Journal of Environmental Chemical Engineering, 3(4), p.3024 - 3029, 2015/12
Humic substances are natural organic matters with heterogeneity in the size distribution and composition of there functional groups. In this study, the effects of organic acid, which are often used as pH buffering agents, on the size of a humic substance and size-dependent binding of metal ions on the humic substance were investigated by flow-field flow fractionation (Fl-FFF). Comparing the three organic acids, Tris, MES, and MOPS, the size of purified Aldrich humic acid (PAHA) changed in the presence of Tris and MES, suggesting that the interaction with these organic acid modulated the molecular structure of PAHA; on the other hand, MOPS hardly affect the size of PAHA. Size-dependent binding of europium (Eu ) and uranium (UO
) and uranium (UO
 ) were also studied by Fl-FFF in the presence of MOPS. The binding of these metal ions were not homogeneous with respect to the size of PAHA; they exhibited high affinity to PAHA fractions with 5 nm hydrodynamic diameter.
) were also studied by Fl-FFF in the presence of MOPS. The binding of these metal ions were not homogeneous with respect to the size of PAHA; they exhibited high affinity to PAHA fractions with 5 nm hydrodynamic diameter.
Saito, Takumi; Terashima, Motoki; Aoyagi, Noboru; Nagao, Seiya*; Fujitake, Nobuhide*; Onuki, Toshihiko
Environmental Science; Processes & Impacts, 17(8), p.1386 - 1395, 2015/08
Times Cited Count:9 Percentile:32.12(Chemistry, Analytical)The deep groundwater HSs were different from surface HSs, having high aliphaticities, sulfur contents, and small molecular sizes. The amounts of their acidic functional groups were comparable to or slightly larger than those of surface HSs; however, the magnitude of Cu binding to the deep groundwater HSs was smaller. The NICA-Donnan model attributed this to the binding of Cu
binding to the deep groundwater HSs was smaller. The NICA-Donnan model attributed this to the binding of Cu to chemically homogeneous carboxylic-type sites via mono-dentate coordination at relatively low pH. The binding mode tended to shift to multi-dentate coordination with carboxylic-type and probably more heterogeneous alcoholic hydroxide-type groups at higher pH. This study shows the particularity of the deep groundwater HSs in terms of their physicochemical and ion-binding properties, compared with surface HSs.
to chemically homogeneous carboxylic-type sites via mono-dentate coordination at relatively low pH. The binding mode tended to shift to multi-dentate coordination with carboxylic-type and probably more heterogeneous alcoholic hydroxide-type groups at higher pH. This study shows the particularity of the deep groundwater HSs in terms of their physicochemical and ion-binding properties, compared with surface HSs.
Ozaki, Takuo; Suzuki, Yoshinori*; Nankawa, Takuya; Yoshida, Takahiro; Onuki, Toshihiko; Kimura, Takaumi; Francis, A. J.*
Journal of Alloys and Compounds, 408-412, p.1334 - 1338, 2006/02
Times Cited Count:47 Percentile:87.11(Chemistry, Physical)We investigated the interactions of Eu(III) with the common soil bacterium Pseudomonas fluorescens and organic ligands, such as malic acid, citric acid, and a siderophore (DFO). Malic acid formed complexes with Eu(III), but degradation of malic acid was observed when the ratio of malic acid to Eu(III) was high. Citric acid formed a stoichiometric complex with Eu(III) that was not degraded by P. fluorescens. Adsorption of Eu(III) from the DFO complex occurred as a free ion dissociated from DFO and not as the Eu(III)-DFO complex. Time-resolved laser-induced fluorescence spectroscopy analysis showed that adsorption of Eu(III) on P. fluorescens was through a multidentate and predominantly inner-spherical coordination.
Kirishima, Akira; Kimura, Takaumi; Tochiyama, Osamu*; Yoshida, Zenko
Radiochimica Acta, 92(12), p.889 - 896, 2005/01
Times Cited Count:24 Percentile:80.96(Chemistry, Inorganic & Nuclear)no abstracts in English
Watanabe, Masayuki; Mirvaliev, R.*; Tachimori, Shoichi; Takeshita, Kenji*; Nakano, Yoshio*; Morikawa, Koshi*; Chikazawa, Takahiro*; Mori, Ryohei*
Solvent Extraction and Ion Exchange, 22(3), p.377 - 390, 2004/06
Times Cited Count:33 Percentile:66.35(Chemistry, Multidisciplinary)The synergistic extraction with N,N,N',N'-tetrakis(2-methylpyridyl)-ethylenediamine (TPEN) and di(2-ethylhexyl)phosphoric acid (D2EHPA) demonstrates a good selectivity for Am(III) over macro amount of Ln(III) in 1-octanol. The maximum apparent separation factor, which is defined as the ratio of the distribution ratio of Am(III) to that of Eu(III), is ca. 80 while the molar fraction between D2EHPA and TPEN is 2.0. This ratio is corresponding to the result of the spectrophotometric titration, which indicates that TPEN and D2EHPA form an aggregate in 1-octanol and two D2EHPA molecules coordinate to Eu(III) TPEN complex, [Eu(TPEN)] . In the present study, the association of D2EHPA and TPEN is one of the most important factors for the synergistic extraction and the complexation of TPEN and D2EHPA with Eu(III).
. In the present study, the association of D2EHPA and TPEN is one of the most important factors for the synergistic extraction and the complexation of TPEN and D2EHPA with Eu(III).
Watanabe, Masayuki; Nankawa, Takuya*; Yamada, Teppei*; Kimura, Takaumi; Namiki, Kosuke*; Murata, Masaki*; Nishihara, Hiroshi*; Tachimori, Shoichi
Inorganic Chemistry, 42(22), p.6977 - 6979, 2003/11
Times Cited Count:34 Percentile:75.17(Chemistry, Inorganic & Nuclear)A tripodal ligand, tris(2-pyridyl)carbinol affords a novel tetradentate coordination mode in homodinuclear lanthanide complexes, which exhibit remarkably short distance between the metal ions. Strong fluorescence of Eu(III) and Tb(III) complexes with the ligand demonstrate that the ligand has a suitable excited state for energy transfer from the ligand to Eu(III) and Tb(III) center.
Tochiyama, Osamu*
JNC TJ8400 2000-044, 53 Pages, 2000/02
To estimate the polyelectrolyte effect and the effect of the heterogeneous composition of humic acids, the complex formation constants of Eu(III) and Ca(II) with Aldrich humic acid and polyacrylic acid were obtained, for Eu(10 to 10
to 10 M) by solvent extraction with TTA and TBP in xylene, for Ca (10
M) by solvent extraction with TTA and TBP in xylene, for Ca (10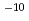 M) with TTA and TOPO in cyclohexane and for Ca(10
M) with TTA and TOPO in cyclohexane and for Ca(10 M) by using ion-selective electrode. By defining the apparent formation as
M) by using ion-selective electrode. By defining the apparent formation as  = [MR
= [MR ]/([M][R]), where [R] denotes the concentration of dissociated functional group, [M] and [MR
]/([M][R]), where [R] denotes the concentration of dissociated functional group, [M] and [MR ] denote the concentration of free and bound metal ion and pcH is defined as-log[H], the values of log
] denote the concentration of free and bound metal ion and pcH is defined as-log[H], the values of log have been obtained at pcH 4.8 - 5.5 in 0.1 - 1.0M NaClO
have been obtained at pcH 4.8 - 5.5 in 0.1 - 1.0M NaClO and NaCl. Log
and NaCl. Log of Eu-humate varied from 5.0 to 9.3 and that of Ca-humate from 2.0 to 3.4..For both humate and polyacrylate, log
of Eu-humate varied from 5.0 to 9.3 and that of Ca-humate from 2.0 to 3.4..For both humate and polyacrylate, log increased with pcH or with the degree of dissociation. The increase in the ionic strength O.1 to 1.0 M decreased the log
increased with pcH or with the degree of dissociation. The increase in the ionic strength O.1 to 1.0 M decreased the log , the decrease in log
, the decrease in log of Eu(III)-humate is 1.6, that of Eu(III), polyacrylate 0.7, that of Ca(II)-humate 1.9 and that of Ca(II)-polyacrylate 1.2. While the increase in the metal ion produced no effect on log
of Eu(III)-humate is 1.6, that of Eu(III), polyacrylate 0.7, that of Ca(II)-humate 1.9 and that of Ca(II)-polyacrylate 1.2. While the increase in the metal ion produced no effect on log of polyacrylate, log
of polyacrylate, log of humate decreased. Depending on the concentration of Eu(III), the coexistence of Ca(II) reduced log
of humate decreased. Depending on the concentration of Eu(III), the coexistence of Ca(II) reduced log  of humate by 0 to 0.8. The dependence of log
of humate by 0 to 0.8. The dependence of log of humate on the metal ion concentration suggests the coexistence of strong and weak binding sites in the hmnic acid.
of humate on the metal ion concentration suggests the coexistence of strong and weak binding sites in the hmnic acid.
Takahashi, Yoshio*; ; Kimura, Takaumi; Meguro, Yoshihiro;
Materials Research Society Symposium Proceedings, Vol.353, 0, p.189 - 196, 1995/00
no abstracts in English
Terashima, Motoki; Saito, Takumi*; Ishii, Tomoko*; Ito, Miki*; Akagi, Yosuke*; Tachi, Yukio
no journal, ,
no abstracts in English
Terashima, Motoki; Saito, Takumi*; Ito, Miki*; Akagi, Yosuke*; Tachi, Yukio
no journal, ,
no abstracts in English
 binding by dissolved organic matters in groundwater
binding by dissolved organic matters in groundwaterTerashima, Motoki; Saito, Takumi*; Akagi, Yosuke*; Endo, Takashi*
no journal, ,
no abstracts in English
Kimuro, Shingo; Iwata, Hajime; Eguchi, Ayano; Nishikawa, Yoshiaki*; Tachi, Yukio
no journal, ,
no abstracts in English
Sato, Hayato*; Toda, Kanako*; Beppu, Hikari*; Amano, Yuki; Miyakawa, Kazuya; Saito, Takumi*
no journal, ,
no abstracts in English
Kobayashi, Taishi*; Sasaki, Takayuki*; Kitamura, Akira
no journal, ,
Solubility of zirconium was measured under gluconate conditions. Gluconate is known as an analogous materials to isosaccharinate which is an organic substance degraded from cellulose. An attempt was applied to obtain an equilibrium constant for zirconium-gloconate systems using the zirconium solubility values as a function of hydrogen and gluconate concentrations.
 -isosaccharinic acid, 2
-isosaccharinic acid, 2Iwata, Hajime; Kimuro, Shingo; Kitamura, Akira; Miyabe, Shunsuke*; Maeno, Mamiko*; Tanaka, Takeru*; Hieda, Manami*
no journal, ,
The effect of  -isosaccharinic acid (ISA) on the solubility of palladium (Pd) in alkaline aqueous solution (pH 8.5, 10.0) was investigated by solubility experiments. The Pd concentration increased with the increase of ISA concentration. The equilibrium constant was calculated from the solubility data obtained in this study.
-isosaccharinic acid (ISA) on the solubility of palladium (Pd) in alkaline aqueous solution (pH 8.5, 10.0) was investigated by solubility experiments. The Pd concentration increased with the increase of ISA concentration. The equilibrium constant was calculated from the solubility data obtained in this study.
Kobayashi, Taishi*; Sasaki, Takayuki*; Kitamura, Akira
no journal, ,
Solubility of Zr in the presence of isosaccharinic acid (ISA) was determined under various pH and ISA concentration ranges. Major aqueous species of Zr-ISA system were estimated and effects of ISA on Zr solubility were discussed based on thermodynamic considerations.